Oxidation
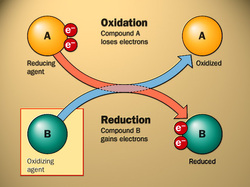
Oxidation=A chemical reaction in which oxygen reacts with some other substance and in which ions, atoms, or molecules lose electrons.
During oxidation, an atom loses electrons and becomes a positively charged ion. (An ion is an atom or a group of atoms that carries an electrical charge, either positive or negative.) Metal atoms tend to undergo oxidation easily.
During oxidation, an atom loses electrons and becomes a positively charged ion. (An ion is an atom or a group of atoms that carries an electrical charge, either positive or negative.) Metal atoms tend to undergo oxidation easily.
